Biobank Ireland Trust was founded in 2004 by Ciaran Flanagan and Professor Eoin Gaffney. A Vodafone Ireland Foundation “World of Difference” award in August 2008 enabled Blanaid Mee to work with Biobank Ireland Trust. With Eoin Gaffney she set up the St James’s Hospital Cancer Biobank in the Department of Histopathology. The new biobank adopted the same honest broker (neutral minders of the samples) policy and procedures kindly provided by the Beaumont Hospital / RCSI Biobank, and the Irish Biobank Network effectively began in these two hospitals. Cork and Galway University Hospitals have adopted the same policies for consent and sample access. Funding for Biobank Ireland Trust has not been obtained from government to date.
Four out of ten people will be diagnosed with cancer during their lifetimes. The development of more effective, targeted treatments and tests for cancer depend on an increased understanding of the molecular mechanisms involved in the initiation of each patient’s cancer, its spread, and its response or resistance to treatment. Large research studies using quality tissue from patients with cancer collected and stored in biobanks are needed to correlate biology with clinical outcome. This is the rationale for an Irish Biobank Network.
To promote among all stakeholders the development of a network of hospital biobanks using a common database to collect, store and share samples donated by patients for approved cancer research projects.
To facilitate (inter)national basic and translational research collaborations and clinical trials that may lead to precise, less toxic treatments and a better prognosis.
To provide an equitable sample access and withdrawal process for all researchers with ethically and scientifically approved projects via a web portal (online catalogue of samples and coded patient data).
To develop with the Faculty of Pathology and the Department of Health a legal mechanism to enable use of archival FFPE tissue and data in research on cancer and other diseases in the public interest.
To engage with the government, patients and members of the public on the importance of a biobanking infrastructure to manage both patient-donated and archival tissue samples for cancer research to benefit patient care, industry, and the economy.
To have government contribute to sustainable funding of co-ordinated biobanking in Ireland as standard of cancer care.
STRATEGIC OBJECTIVES (2022)
- Network of biobanks
- Public / patient engagement
- Fair access to research samples and data: shared BIMS, web portal
- ISO and GDPR; plan for accreditation
- Freezer with TCD and SJH
- Fundraising
Biobank Ireland Trust (Charities Regulator No. 20057176) is a company limited by guarantee, managed by a Board of Trustees.
Board of Trustees: Ciaran Flanagan, Eoin Gaffney, Richard Flavin.
5 New Trustees are about to join the Board.
Chief Scientific Advisor: B Mee
Biobank Ireland complies with The Charities Regulator’s Governance Code – reviewing its charitable purpose, strategic plan, budgets, procedures, stakeholder accountability, legal requirements and accounts at quarterly Board meetings. The Board oversees communications, diverse stakeholder engagement including patient groups and the public, industry, hospitals, universities, government, and fundraising.
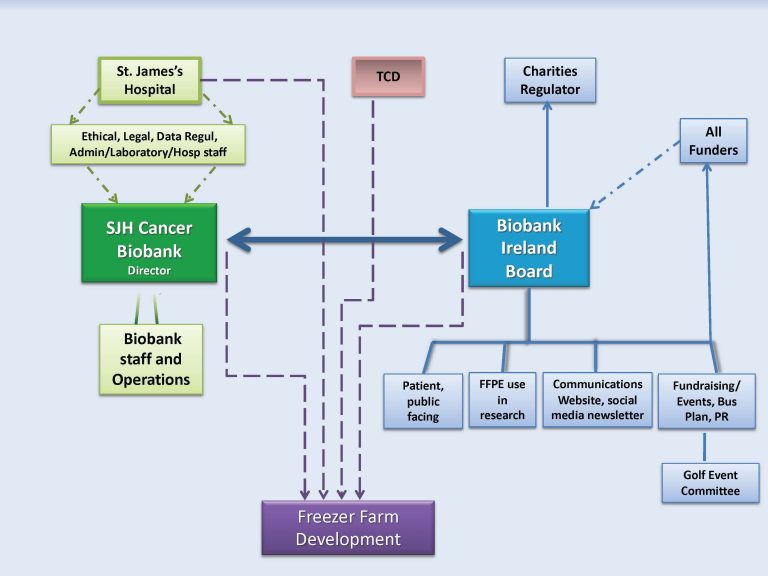
Biobank Ireland Trust’s Board of Trustees and St James’s Hospital have joint responsibility for governance of the St James’s Hospital Cancer Biobank.
On a day to day basis, OPERATIONS (biobanking, BIMS development, procedures, policies, equipment needs, and project applications) are managed by the Biobank director and Biobank staff, guided by the Operations Executive and Chief Medical Scientist (Department of Histopathology), who liaise with the Research and Innovation Office, Research and Ethics Committee, Data Protection Officer, and Risk and Legal Office. St James’s Hospital is the Data Controller for the BIMS and is also involved through the Central Pathology Laboratory directorate. Operational meetings are held weekly.
Biobank Ireland Trust’s main role within the SJHCB is in STRATEGIC PLANNING. Biobank Ireland provides funds for the BIMS, and essential equipment, projects and staff and discusses progress and strategy at monthly SJHCB / Biobank Ireland meetings.
Biobank Ireland promoted the concept of a Freezer for the St James’s-TCD campus for all researchers investigating cancer and other diseases. This exciting development is at the pre-planning stages: bi-monthly meetings are held between Biobank Ireland and senior representatives from St James’s Hospital and Trinity College.
| Details | Biobank Ireland Trust |
| Business Name | Cancer Research Progress & Support Ltd. |
| Trading Name | Biobank Ireland Trust |
| Company Number | 394406 |
| Corporation Tax Number | 64144060 |
| Charity Regulatory Authority Website Number | 20057176 |
| Charity Number | CHY 16085 |
| Bank Account Details | AIB 1 Lower Baggot Street |
| Sort Code | 931012 |
| BIC | AIBKIE2D |
| IBAN | IE20 AIBK931012062930 31 |
Biobank Ireland receives funds from several sectors – from industry, business, philanthropists, and from fundraising events but not from government. Multisector funding will be needed to develop the Irish Biobank Network, which is most likely to develop by formal institutional biobanking collaboration (as in Canada and Israel) rather than by organic growth.
Biobank Ireland chiefly supports the St James’s Hospital Cancer Biobank (see Governance) and together with partners SJH and TCD is developing a large centralised biobank facility for all researchers on the SJH-TCD campus and for network and industry users. This will reduce duplication of effort and will improve research efficiency and ease of collaboration.
The first stage of planning application has been approved. Funding this development will require grants and coordinated fundraising by the three partners.


